Why Dehydration Is Required
Natural gas from wells invariably contains water vapour, the quantity of which depends on reservoir pressure, temperature, and gas composition. Gas also becomes saturated with water in the Acid Gas Removal Unit (AGRU) of a gas processing facility, where it comes into contact with the aqueous solution of an amine solvent — such as Methyldiethanolamine (MDEA) — used to remove H2S and CO2 from the raw gas.
Water must be removed from natural gas because its condensation or reaction with hydrocarbons causes several serious operational problems:
- Pipeline corrosion — in the presence of CO2 and/or H2S, liquid water forms carbonic acid and bisulphide species that aggressively corrode carbon steel pipelines and equipment
- Hydrate formation — water and light hydrocarbons (C1–C4) combine at elevated pressure and low temperature to form solid crystalline hydrate plugs, which can block pipelines, valves, and instrumentation
- Slug formation — liquid water accumulation in low points of gathering and transmission pipelines causes damaging slugging and flow assurance problems ²
- Reduction in heating value — water vapour dilutes the gas stream, increasing volume while reducing calorific value per unit volume
- Plugging of cryogenic equipment — ice formation in turboexpanders, cold boxes, and LNG heat exchangers requires water dewpoint specifications of typically less than −100°C, achievable only with molecular sieves
Glycol Properties & Selection
The most widely used desiccants for absorption-based gas dehydration are Diethylene Glycol (DEG) and Triethylene Glycol (TEG). Both compounds contain hydroxyl groups (–OH) and ether functional groups (–CH2–O–CH2–) in their molecular structure. These polar groups form strong hydrogen bonds with water molecules, explaining their exceptional hygroscopicity and desiccant properties.
Because glycols have molecular weights considerably greater than water, they boil at significantly higher temperatures and can be readily regenerated from the water-glycol solution by distillation. However, both DEG and TEG are subject to thermal degradation at elevated temperatures; regeneration must therefore be carried out below their respective decomposition onset temperatures:
| Glycol |
Decomposition Onset |
Normal Boiling Point |
| DEG |
164°C (328°F) |
245°C (473°F) |
| TEG |
207°C (404°F) |
287°C (549°F) |
Since their normal boiling points exceed their decomposition temperatures, atmospheric pressure distillation alone is insufficient to achieve deep regeneration. In practice, stripping gas (dry fuel gas or nitrogen) is injected into the reboiler or a dedicated stripping column to enhance water removal at safe temperatures.
TEG is preferred for gas dehydration service because its higher decomposition temperature allows more aggressive regeneration, achieving lean glycol concentrations of 99.5–99.99 wt% and water dewpoints of −40°C to −70°C under standard conditions. DEG is preferred for hydrate inhibition service (injection into wellheads and flowlines) where lean glycol concentrations of 85–95 wt% are sufficient and lower regeneration temperatures are advantageous.
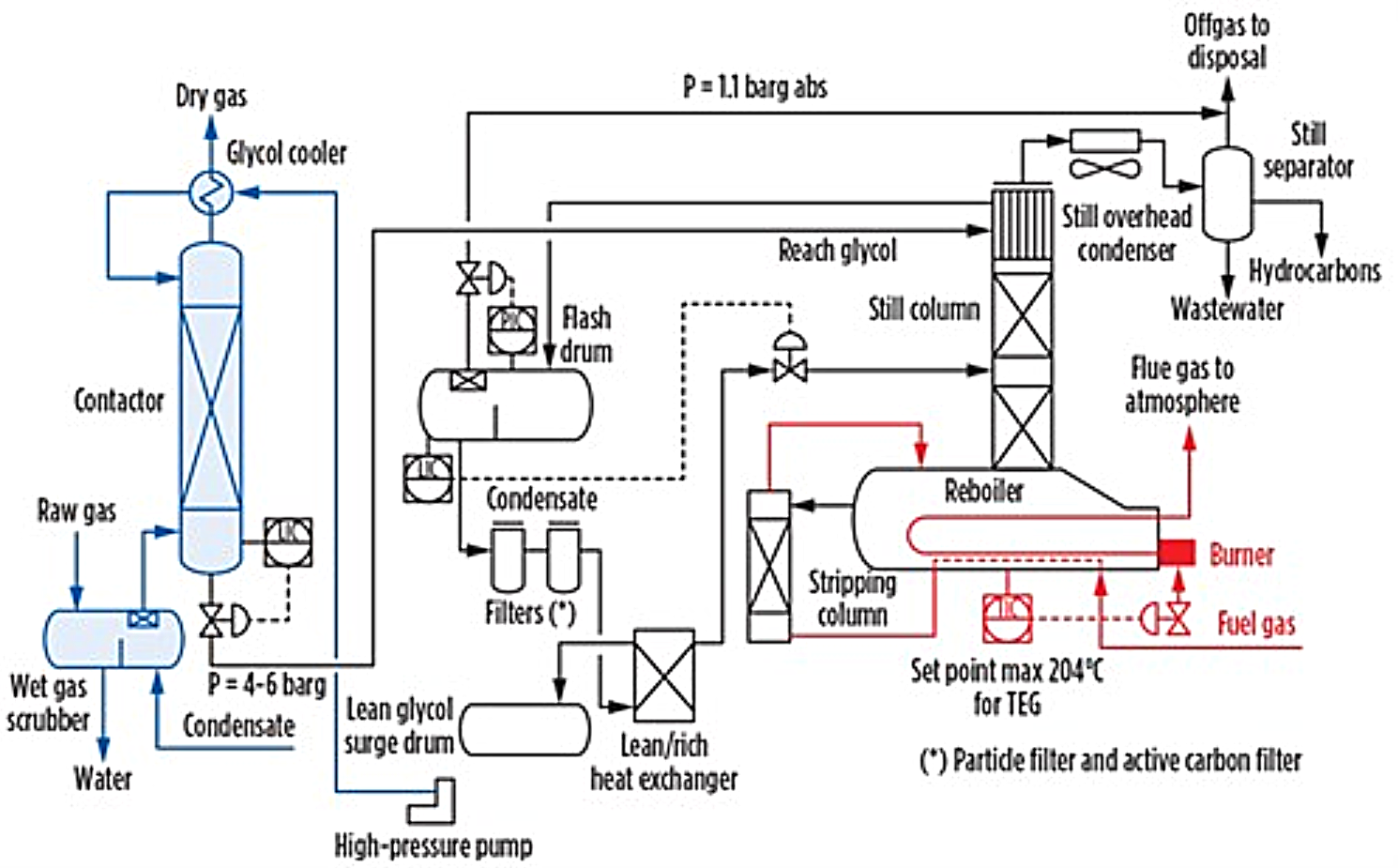
Process Description
Glycol dehydration is a regenerative absorption process operating continuously in two integrated sections: absorption and regeneration.
Absorption Section
Raw wet gas first passes through an inlet gas scrubber / separator upstream of the contactor to knock out free liquid water, condensate, and entrained solids. The scrubbed gas enters the base of a high-pressure contactor column, where it flows upward in countercurrent contact with a descending stream of lean glycol. Water vapour is selectively absorbed by the glycol, and dried gas exits from the top of the contactor through a mist eliminator.
Contactor internals may be valve trays, random packing, or structured packing. Structured packing is increasingly preferred for new designs due to its higher specific gas capacity, lower glycol entrainment, and smaller required column diameter.
The rich glycol withdrawn from the contactor sump passes through a level control valve and is routed to a glycol flash drum, where dissolved light hydrocarbons and inert gases are separated and recovered as fuel gas.
Regeneration Section
After flash separation, the rich glycol undergoes particulate and activated carbon filtration to remove solid contaminants and BTEX compounds, followed by pre-heating in a lean/rich glycol heat exchanger before entering the still column.
In the still column, water is stripped from the glycol by rising vapours generated in the glycol reboiler operating at approximately 190–205°C for TEG. The regenerated lean glycol is cooled through the lean/rich heat exchanger and a trim cooler before being pumped back to the contactor by the glycol circulation pump.
Stripping Enhancement (Optional)
Where deeper dehydration is required, stripping gas (dry fuel gas or nitrogen) introduced below the reboiler increases lean glycol purity from ~99.0–99.5 wt% to >99.9 wt%, achieving water dewpoints approaching −80°C.
Key Design Parameters
| Parameter |
Typical Value |
| Operating pressure (contactor) |
30–100 bara |
| Lean TEG concentration |
99.0–99.99 wt% |
| TEG circulation rate |
15–40 litres TEG per kg H2O removed |
| Reboiler temperature (TEG) |
190–205°C |
| Achievable water dewpoint |
−40°C to −70°C (standard); to −80°C with stripping |
| TEG losses |
0.01–0.1 kg/MMscf treated gas |
Limitations
Glycol dehydration cannot achieve the water dewpoint specifications required for cryogenic LNG plants (<−100°C, typically <0.1 ppm water). For these applications, molecular sieve dehydration (Temperature Swing Adsorption, TSA) is mandatory, either alone or downstream of a glycol unit used as a bulk water removal pre-treatment step.
References
- Gas Processing News (December 2020). The Natural Gas Dehydration Process
- Kohl A.L. & Nielsen R.B. (Aug 28, 1997). Gas Purification, 5th edition. Gulf Publishing Company, Houston, TX. Chapter 12: Dehydration of Natural Gas
- Sloan E.D. & Koh C.A. (2007). Clathrate Hydrates of Natural Gases, 3rd edition. CRC Press, Boca Raton, FL
- GPSA Engineering Data Book, 14th edition (2017). Gas Processors Suppliers Association, Tulsa, OK. Section 20: Dehydration
- Campbell J.M. (2004). Gas Conditioning and Processing, Vol. 2: The Equipment Modules, 8th edition. Campbell Petroleum Series, Norman, OK. Chapter 13: Glycol Dehydration