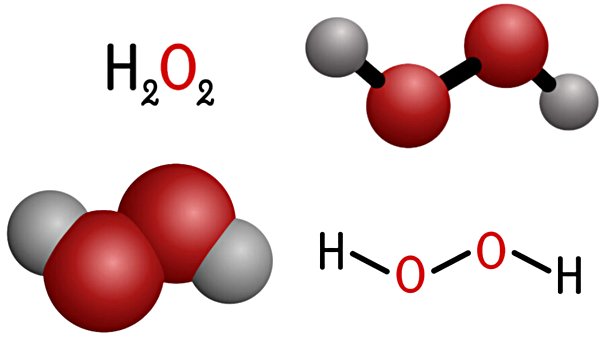
Hydrogen peroxide (H₂O₂) is a colorless, slightly viscous liquid with the molecular formula H₂O₂ and a molecular weight of 34.01 g/mol. In pure form it is a pale blue liquid, but at commercial concentrations it appears colorless. It is structurally the simplest peroxide — a compound containing an oxygen–oxygen single bond — and can be described as water with one additional oxygen atom. The molecule is asymmetric and highly polarized, with a strong tendency to form hydrogen bond networks, giving it greater viscosity than water.
Physical and Chemical Properties
| Property |
Value |
| Molecular formula |
H₂O₂ |
| Molecular weight |
34.01 g/mol |
| Appearance |
Colorless to pale blue liquid |
| Melting point |
−0.43°C (pure, 100%) |
| Boiling point |
150.2°C (pure, 100%) |
| Density |
1.45 g/cm³ (100%); 1.11 g/cm³ (30%) |
| Miscibility |
Fully miscible with water |
| Decomposition |
H₂O₂ → H₂O + ½ O₂ (exothermic) |
Hydrogen peroxide is thermodynamically unstable and decomposes spontaneously to water and oxygen, releasing heat. The rate of decomposition is accelerated by heat, light, alkaline conditions, and contact with metal ions (Fe, Cu, Mn, Cr, Ni) or organic matter. For this reason, commercial products are stabilized with small quantities of acidic stabilizers (typically acetanilide or phosphonate compounds) and stored in opaque containers at weakly acidic pH. At concentrations above ~70 wt%, hydrogen peroxide can decompose explosively when heated and is classified as a strong oxidizer that can cause spontaneous combustion on contact with organic materials.
Commercial Grades and Concentrations
Hydrogen peroxide is commercially produced and traded as aqueous solutions across a range of concentrations:
| Grade |
Concentration |
Principal use |
| Consumer / pharmaceutical |
3–9 wt% |
Antiseptic, wound care,
cosmetics |
| Technical |
30–35 wt% |
General industrial
bleaching |
| Electronic grade |
30–35 wt% |
Semiconductor cleaning
(ultra-pure) |
| Textile / paper |
35–50 wt% |
Pulp and textile
bleaching |
| Chemical synthesis |
50–70 wt% |
Propylene oxide (HPPO),
caprolactam, epoxidation |
| Propellant grade |
85–98 wt% |
Rocket propellant, monopropellant |
Production
Over 95% of global hydrogen peroxide is produced via the Riedl-Pfleiderer anthraquinone auto-oxidation (AO) process, in which alkylated anthraquinones serve as oxygen carriers in a cyclic hydrogenation-oxidation sequence; the net reaction is H₂ + O₂ → H₂O₂. The largest producers are Solvay, Evonik Industries, BASF, Arkema, and Mitsubishi Gas Chemical. Global production capacity exceeds 6 million tonnes per year (100% basis).
Industrial Applications
Hydrogen peroxide is a versatile oxidizing and bleaching agent used across a broad range of industries:
- Chemical synthesis: epoxidation of propylene to propylene oxide via the HPPO process; synthesis of caprolactam, cyclohexanone oxime, catechol, and peracids
- Pulp and paper: elemental chlorine-free (ECF) and totally chlorine-free (TCF) bleaching of wood pulp
- Textile: bleaching of cotton, linen, and synthetic fibres
- Environmental: wastewater treatment, advanced oxidation processes (AOP), soil remediation
- Electronics: precision cleaning of silicon wafers and semiconductor substrates
- Mining: gold and uranium heap leaching
- Food: sterilization of packaging, bleaching of flour and starch
- Cosmetics and personal care: hair bleaching and tooth whitening
- Aerospace and defense: monopropellant and oxidizer in rocket propulsion systems
Safety and Handling
Hydrogen peroxide is classified as a strong oxidizer (UN 2014 at 8–60%; UN 2015 above 60%) and must be stored away from flammable materials, reducing agents, and transition metal contaminants. Solutions above 8 wt% are corrosive to skin and mucous membranes. Industrial handling requires dedicated stainless steel, aluminium, or HDPE equipment; carbon steel and copper alloys must be avoided as they catalyze decomposition. Concentrated grades (>70 wt%) require explosion-proof equipment and specialized storage.
References
- Wikipedia — Hydrogen peroxide (Page version: Mar 28, 2026)
- Britannica — hydrogen peroxide (Page version: Mar 21, 2026)
- Medical News Today — West M. (Jul 21, 2021). What to know about hydrogen peroxide
- Agency for Toxic Substances and Disease Registry (ATDSR) — Medical Management Guidelines for Hydrogen Peroxide (Accessed Apr 5, 2026)
- Agency for Toxic Substances and Disease Registry (ATDSR) — Hydrogen peroxide (Accessed Apr 5, 2026)
- Alliance Chemical — Taki A. (Jun 3, 2025). The Complete Hydrogen Peroxide Concentration Guide: 3% to 30%
- Evonik — What Is Hydrogen Peroxide? (Accessed Apr 5, 2026)