Direct oxidation routes for propylene oxide production added
- Technology Type
- Epoxidation of Propylene with Peroxides
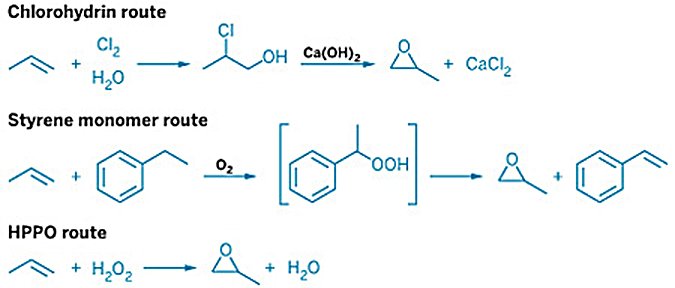
Propylene oxide (PO) is one of the chemical industry's most important intermediates, produced at over 10 million tonnes per year globally and consumed primarily as the precursor to polyurethane polyols, propylene glycol, and glycol ethers. The historical chlorohydrin process, introduced in the 1950s, was the first industrial PO-only route but generated large salt effluents and required integrated chlorine infrastructure.
To address these limitations, epoxidation routes — in which propylene is epoxidized using a preformed peroxide oxidant — were developed as cleaner, chlorine-free alternatives from the 1960s onward. These routes are the dominant commercial pathway for PO production worldwide and have displaced the chlorohydrin process for all new capacity since the early 1970s. They can be grouped into two families:
-
Organic hydroperoxide routes: propylene is epoxidized by an organic hydroperoxide derived from a hydrocarbon co-feed — ethylbenzene (SMPO), isobutane (TBA), or cumene (POC). The first two co-produce organic by-products; the cumene route recycles its carrier and produces PO as the sole product.
-
Hydrogen peroxide route (HPPO): propylene is epoxidized with hydrogen peroxide (H2O2) over a TS-1 zeolite catalyst in methanol solvent; water is the only by-product.
Read this detailed 'Epoxidation of Propylene with Peroxides' technology profile here.
#propyleneoxidation #epoxidation #propyleneoxide #hppo #posm #smpo #tba #peroxide #styrenehydroperoxide #coproduct #hydrogenperoxide #cumene #cumenehydroperoxide #hppo #evonik #basf #dow #lyondellbasell #shell #huntsman #sumitomo #thyssenkrupp #uhde