Technology History
The HPPO (Hydrogen Peroxide to Propylene Oxide) process emerged from converging independent development tracks spanning two decades. The TS-1-catalyzed epoxidation of propylene into propylene oxide (PO) with hydrogen peroxide (HP) was first invented by EniChem (Italy) in the early 1980s. BASF independently began exploring hydrogen peroxide-based routes to propylene oxide from the mid-1990s, developing a tubular fixed-bed reactor concept with a proprietary TS-1 catalyst. Dow Chemical acquired its know-how from its 2001 purchase of EniChem's polyurethane business, which included EniChem's TS-1 catalyst-in-suspension approach.
The two companies began examining each other's processes in 2002 and formally entered joint development in 2003, combining BASF's fixed-bed reactor design and Dow's catalyst heritage into a single optimized process. The decisive joint development contributions included optimization of the propylene/H₂O₂/methanol feed ratios, the two-step reactor concept with intermediate PO separation, the buffered feed system, and the energy-integrated extractive distillation. Solvay SA joined as a long-term hydrogen peroxide supply partner for the first plant.
The first world-scale commercial unit — a 300,000 t/y single-train plant — started up smoothly in Q4 2008 at BASF's Verbund site in Antwerp, Belgium. The process received prominent industry recognition: the IChemE Award (2009), the Kirkpatrick Honour Award (2009), and the U.S. EPA Presidential Green Chemistry Challenge Award (2010). Patent activity by BASF and Dow on this process remains active as recently as 2024.
Technology Summary and Competitive Context
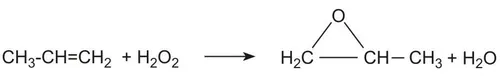
The BASF-Dow HPPO process produces propylene oxide (PO) by direct liquid-phase epoxidation of propylene with hydrogen peroxide in methanol solvent over a proprietary TS-1 zeolite catalyst. The only raw materials are propylene and hydrogen peroxide; the sole stoichiometric by-product is water (~300 kg per tonne PO). This eliminates the co-product management burden that characterizes all older PO routes.
| Route |
Co-product per t PO |
Infrastructure burden |
| Chlorohydrin (CHPO) |
~2.1 t CaCl₂ + 40 t saline water |
Chlor-alkali plant required |
| POSM (ethylbenzene hydroperoxide) |
~2.2 t styrene |
Styrene market dependency |
| PO/TBA (isobutane route) |
~2.4 t t-butanol / MTBE |
MTBE market dependency |
| Cumene HP (Sumitomo) |
~1.5 t cumyl alcohol (recycled) |
Internal recycle loop |
| HPPO (BASF-Dow) |
~0.3 t water only |
None |
Versus conventional routes, HPPO reduces wastewater generation by up to 80% and energy consumption by 35%, with an ISBL capital investment 10–15% lower than POSM-type hydroperoxidation processes. Propylene utilization is also more efficient: the overall propylene excess required is significantly below 50% compared to organic hydroperoxide routes.
Chemistry and Mechanism
Main reaction:
[Conditions: TS-1, MeOH, <90°C, 30 bar] [Rate constants k0 (uncatalyzed H2O2 consumption and k1 (TS-1 catalysed reaction)]
The catalytic mechanism involves a hydroperoxotitanium intermediate (Ti–OOH) formed at the tetrahedral Ti⁴⁺ sites of the TS-1 lattice, stabilized by methanol coordination. This species electrophilically delivers oxygen to the electron-rich propylene double bond, generating PO and regenerating the Ti–OH site. Methanol is not merely an inert solvent: it participates directly in catalyst activation and phase homogenization, preventing liquid-phase separation between the hydrophobic propylene and the aqueous H₂O₂.
Key side reactions (all to be minimized):
PO + MeOH → methoxypropanols (1-MeO-2-PrOH and 2-MeO-1-PrOH) [Rate constant k2]
H2O2 → H2O + ½O2 (decomposition — yield loss and O₂ safety hazard) [Rate constant k3]
The rate constants satisfy k0 ≈ k1 ≫ k2, k3, k4 at operating temperature, enabling high PO formation rate relative to loss reactions. Increasing H₂O₂ conversion by raising temperature or extending residence time increases PO concentration, which in turn accelerates ring-opening reactions — creating a fundamental kinetic trade-off that motivates the two-step reactor design.
Feed buffering: The reactor feed is conditioned with a potassium phosphate buffer system, providing K⁺ cations at 110–190 micromol per mol H₂O₂ in the combined feed. This suppresses acid-catalyzed PO ring-opening and controls catalyst deactivation by neutralizing trace acidic by-products. The HP/methanol mixture is held for a minimum contact time of ~8 seconds and filtered before entering the epoxidation zone to ensure feed homogeneity.
Optimized feed ratios (from joint pilot development):
- Propylene : H₂O₂ molar ratio: 1.1–1.5 mol/mol (optimal range; below 1.0 causes biphasic liquid separation; above 1.6 inflates recycle costs)
- Methanol : H₂O₂ weight ratio: 5:1 to 15:1 (minimum sufficient to maintain single liquid phase)
- Combined feed H₂O₂ concentration: <10 wt% in the reactor mixture
- Propylene : H₂O₂ weight ratio in feed: 1:1 to 5:1
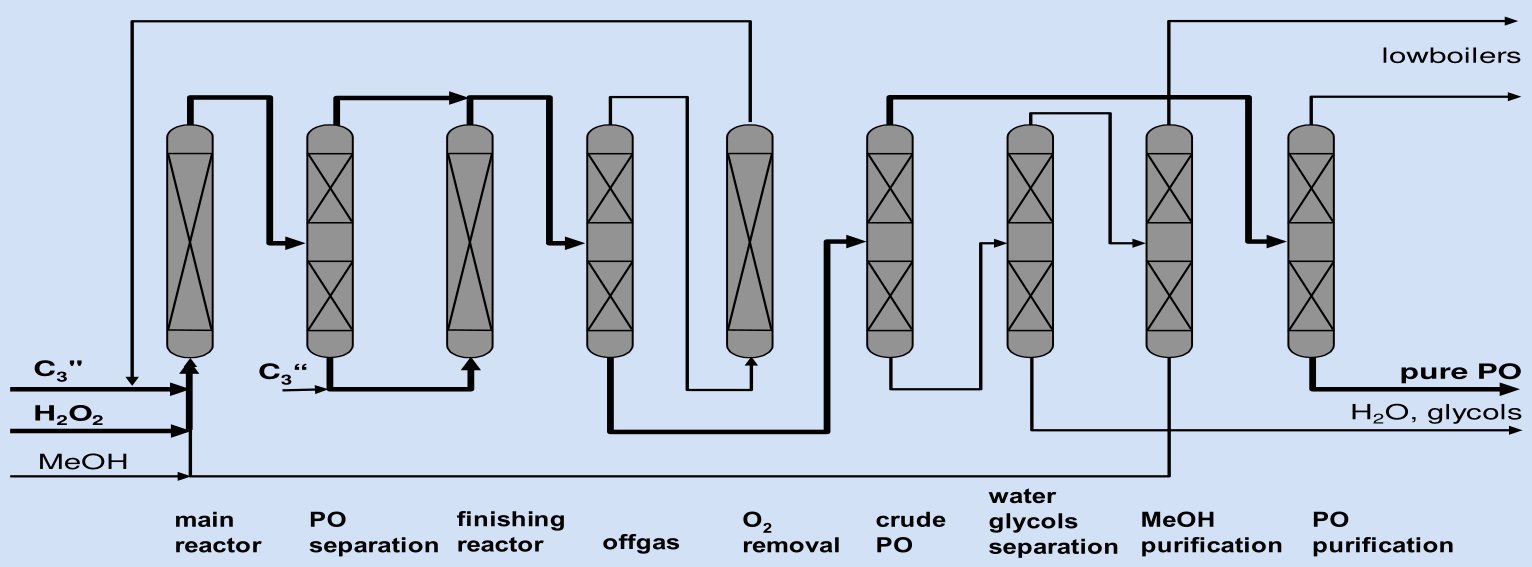
Step-by-Step Process Description
The complete process comprises eight sections:
Step 1 — Main Epoxidation Reactor
Fresh propylene, aqueous H₂O₂ (30–70 wt%), and recycled methanol are combined, buffered with K₂HPO₄, and fed to a tubular fixed-bed reactor loaded with the proprietary TS-1 Ti-zeolite catalyst. The reactor operates in liquid phase at below 90°C and ~30 bar; cooling jackets or internal heat exchangers maintain isothermal conditions across the catalyst beds. Nitrogen is injected into the vapor space as a ballast gas to prevent formation of flammable propylene/oxygen mixtures, the oxygen arising from trace H₂O₂ decomposition. The design conversion target for this first reactor is ~90% H₂O₂ conversion.
Step 2 — PO Separation (Inter-stage Distillation)
The first reactor effluent is depressurized to near-atmospheric pressure in a flash/distillation column operating at a bottom temperature of ~69°C. PO is distilled overhead together with unreacted propylene and a fraction of methanol. The bottom stream — containing residual H₂O₂ (~10% of original), methanol, water, and high-boiling by-products — feeds directly to the finishing reactor. This intermediate PO removal step is the architectural key of the two-step concept: by reducing PO concentration in the liquid phase before the second reactor, the consecutive ring-opening reactions (k₂, k₃ pathways) are kinetically suppressed, allowing the finishing reactor to drive H₂O₂ to near-complete conversion without sacrificing selectivity.
Step 3 — Finishing Reactor
Fresh propylene is added to the inter-stage bottom stream, and the mixture enters the finishing reactor operating under the same temperature and pressure conditions as the main reactor. The finishing reactor raises overall H₂O₂ conversion from ~90% to above 99%, achieving a second-step PO selectivity of 96% based on H₂O₂.
Step 4 — Off-gas Treatment and Light-Boiler Separation
The combined overhead from both PO separation stages contains: propylene (unreacted), propane (introduced with chemical-grade propylene), nitrogen ballast, oxygen from H₂O₂ decomposition, and volatile by-products (formaldehyde, acetaldehyde). This off-gas stream is compressed to ~16 atm, cooled to 35°C, and fed to a pressurized stripping column, yielding:
- Bottoms: propylene-rich stream (with any propane), sent to Step 5
- Overhead: low-hydrocarbon off-gas containing N₂, O₂, and volatile by-products, purged from the system
Step 5 — Oxygen Removal and Propylene Recycle
Before the propylene-rich stream from Step 4 can be recycled to the main reactor, oxygen must be removed to prevent flammable mixtures forming in the reactor vapor space. This is accomplished by catalytic reduction over a tin/noble metal catalyst: oxygen reacts selectively with hydrogen (H₂:O₂ molar ratio ≥5) to produce water, at conditions that do not affect propylene. The de-oxygenated propylene stream is recycled directly to the main reactor. If chemical-grade propylene is used as feedstock, a propane bleed is taken at this step to prevent inert alkane build-up in the recycle loop.
Step 6 — Crude PO Purification (Extractive Distillation)
PO and methanol form a near-azeotropic mixture that cannot be separated by simple distillation. The BASF-Dow process employs sub-atmospheric extractive distillation:
- Operating pressure: 300–750 mbar
- Column bottom temperature: 40–70°C
- Feed composition to column: 5–50 wt% PO, 50–85 wt% methanol
- Extractant: water (or propylene glycol as alternative) introduced at the column top
- Overhead product: pure PO (target ≤100 ppm methanol)
- Bottom stream: methanol + water, forwarded to Step 7
The overhead PO vapor is compressed and the condensation heat is returned to the vaporizer of the same extractive column, providing significant energy integration. The reflux-to-distillate ratio is kept ≤4.
Step 7 — Methanol Purification and Recycle
The methanol-water bottom stream from Step 6 is processed in a methanol purification unit where methanol is separated by distillation and recycled to the reactor feed. A small quantity of methyl formate (formed from formaldehyde + methanol during the reaction) co-elutes and must be removed in this step to deliver a clean methanol recycle stream. The bottoms from methanol purification — water with trace glycols and methoxypropanols — is tested analytically before discharge to the wastewater treatment unit. The methanol make-up requirement is only ~14 kg per tonne PO produced, reflecting near-complete recycle efficiency.
Step 8 — PO Final Purification
Crude PO from the extractive distillation overhead is further refined in a final distillation column to achieve polymer-grade or chemical-grade PO purity (≥99.8 wt%). Low-boiling impurities (formaldehyde, acetaldehyde) exit overhead; pure PO is drawn as a side or bottoms stream. The glycols and water remaining after methanol recovery exit the process as the wastewater stream, containing only stoichiometric water (~300 kg/t PO) plus trace by-products.
Process Efficiency
Reactor Performance by Step
| Stage |
H₂O₂
Conversion |
PO Selectivity
(vs. H₂O₂) |
PO Yield
(vs. H₂O₂) |
| Main reactor (Step 1) |
~90% |
~95% |
~85% |
| Finishing reactor (Step 3) |
~96% (2nd step only) |
~96% |
~92% |
| Overall (two-step) |
99.4% |
95–96% |
94–95% |
| Single reactor (reference) |
~98.4% |
~80.3% |
~79% |
The two-step concept delivers a ~15–16 percentage point improvement in PO yield versus a single-reactor design at equivalent H₂O₂ conversion.
Catalyst Performance
The TS-1 catalyst undergoes reversible deactivation due to PO oligomer formation on the active sites, with H₂O₂ conversion declining from ~96% to ~80% after approximately 135 hours of continuous operation at 70°C. Full activity is recovered by calcination in air at 873 K (600°C). In the Evonik-Uhde variant (closely related chemistry), catalyst regeneration can also be performed by washing with methanol at ≥100°C.
Outlet Stream Composition
Representative composition of the liquid stream leaving the epoxidation reactors, after separation of light-boiling compounds:
| Component |
Mass fraction |
| Methanol |
71.97% |
| Water |
17.54% |
| Propylene oxide (PO) |
9.45% |
| Glycol ethers |
0.43% |
| Propylene glycol |
0.05% |
| Formaldehyde |
0.01% |
| Acetaldehyde |
0.03% |
| Unreacted propylene |
0.01% |
| Heavy boilers |
remainder |
Raw Material Consumption (estimate)
| Input |
Consumption |
| Propylene (polymer grade) |
0.778 t |
| H₂O₂ (100% basis, supplied as 40% solution) |
0.750 t |
| Methanol make-up |
0.014 t |
| Water co-product output |
~0.300 t (stoichiometric only) |
Commercial Experience
| Plant |
Location |
Capacity |
Start-up |
Partners / HP supply |
| BASF Verbund site |
Antwerp, Belgium |
300,000 t/y |
Q4 2008 |
BASF + Dow (PO); Solvay 230,000 t/y HP co-located |
| Dow / Siam Cement Group |
Map Ta Phut, Thailand |
390,000 t/y |
2011 |
Dow + SCG (PO);
Solvay 330,000 t/y HP co-located |
| Sadara Chemical Co. |
Jubail II, Saudi Arabia |
~390,000 t/y |
~2015 |
Dow + Saudi Aramco (PO);
Saudi HP Co. (Solvay-Sadara JV) HP co-located |
A consistent co-location model characterizes all three BASF-Dow HPPO deployments: Solvay constructs and operates a dedicated world-scale hydroperoxide (HP) plant adjacent to each PO unit, eliminating HP transport logistics and the associated safety constraints of shipping concentrated hydrogen peroxide over long distances. The Antwerp plant remains the only 300 kta single-train unit and is fully integrated into BASF's Verbund chemical complex. The Thailand plant, at 390 kta, was supplied with propylene from a Dow/Siam Cement Group liquids cracker built concurrently at the same industrial site.
References
- Sustainable Chemistry — Lew D. (Dec 4, 2023) From the Dream Reaction to the Real Process the Implemented HPPO Process
- The Essential Chemical Industry - online — (Jan 6, 2017). Propene (Propylene)
- Bassler P., Weidenbach M. and Goebbel H., (2010), The new HPPO Process for Propylene Oxide: From Joint Development to Worldscale Production. Chemical Engineering Transactions, 21, 571-576. DOI: 10.3303/CET1021096
- BASF SE / Dow Chemical Co —Teles J.H. et al. (Filed: Dec 6, 2011). United States patent US8785670B2: Process for the production of propylene oxide
- BASF SE / Dow Chemical Co. Gobbel H.G. et al. (Filed: Jul 7, 2004). United States patent US20060009648A1: Separation of propylene oxide from a mixture comprising propylene oxide and methanol
- BASF SE. Teles J.H. et al. (Filed: Jul 5, 2001). United States patent US6756503B2: Method for the production of propylene oxide
- BASF SE / Dow Chemical Co. Schindler G.P. et al. (Filed Jun 29, 2005). United States patent US20070004926A1: Process for producing propylene oxide
-
- BASF SE / Dow Global Technologies LLC. Chemical. Parvulescu et al. (Filed: Jun 22, 2022). United States patent US20240294483A1: Process for the preparation of a liquid mixture comprising methanol, water and hydrogen peroxide. Patent US20240294483A1, 2024. (Feed preparation — most recent disclosure)
- Gu et al. (Nov 16, 2018). Validation of the Kinetics of the Hydrogen Peroxide Propene Oxide (HPPO) Process. Industrial & Engineering Chemistry Research, 57(48), 16201–16208. DOI: 10.1021/acs.iecr.8b03233
- Saudi Gazette — (Sep 2, 2013). World-scale hydrogen peroxide plant in Kingdom to open in 2015
- Sadara Chemical Company — (Sep 2, 2013). Press Release: Solvay and Sadara joint venture begins construction of world-scale hydrogen peroxide plant in Saudi Arabia