Technology History
The HF/aluminium hydroxide process for synthetic cryolite production developed directly from the industrial fluorochemical industry in the mid-20th century, as natural cryolite supplies from the Ivigtut deposit in Greenland — the world's sole commercial natural source — began to decline and ultimately became exhausted in 1987. The transition to fully synthetic production was driven by the rapid post-war expansion of primary aluminium smelting capacity, which required a reliable, scalable, and consistent-quality sodium hexafluoroaluminate (cryolite) supply that natural mining could not provide. The HF-based route leveraged existing hydrofluoric acid manufacturing infrastructure — itself based on the reaction of fluorspar with sulphuric acid — and was adopted commercially by European and North American producers from the 1950s onward. It remains the dominant global route today, favoured for producing the highest purity Na₃AlF₆ with the most consistent cryolite ratio (CR = Na:Al molar ratio, target = 3.0) required by aluminium smelters.
Technology Summary
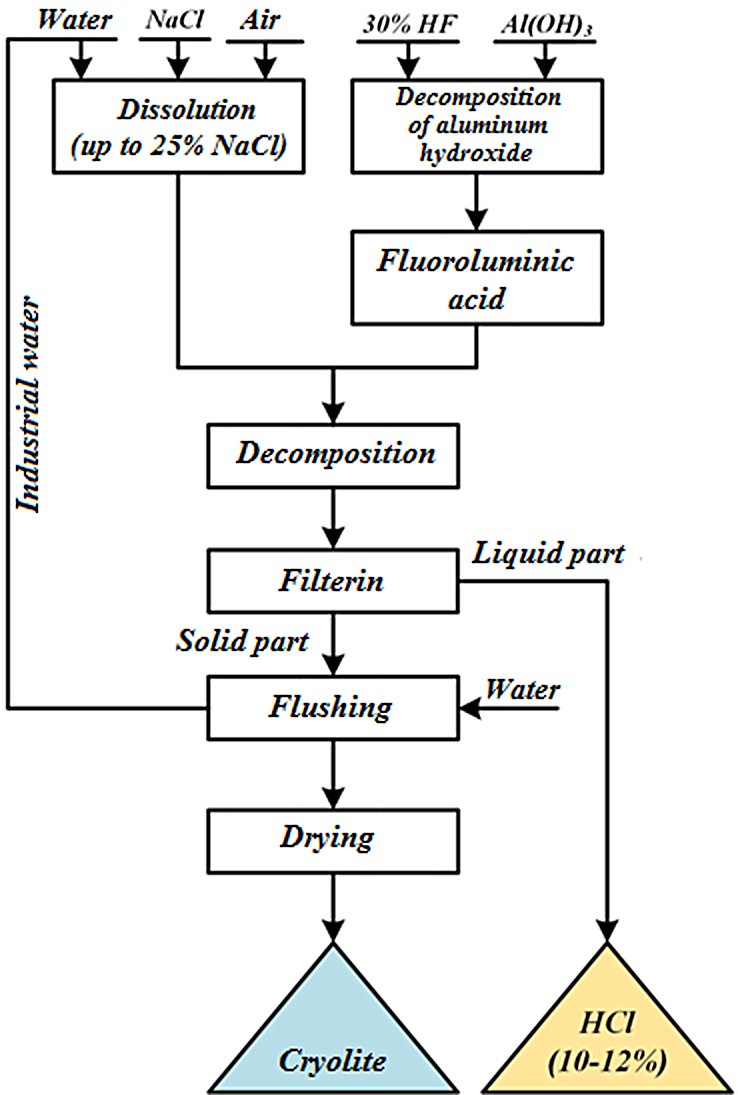
The process is a wet chemical precipitation technology in which hydrofluoric acid reacts with aluminium hydroxide to form an intermediate fluoroaluminic acid solution, which is then converted to sodium hexafluoroaluminate by a double-replacement reaction with a sodium source (sodium chloride or sodium carbonate), followed by solid-liquid separation, washing, and calcination to produce the final granular or milled product.
Chemistry
Step 1 — Complexation (fluoroaluminic acid formation):
Al(OH)3 + 6 HF → H3AlF6 + 3 H2O
This is an acid-base reaction in which HF (weak acid) displaces hydroxide from aluminium, forming the octahedral [AlF₆]³⁻ complex stabilised by three protons. The reaction is exothermic and proceeds readily at ambient temperature.
Step 2 — Ion exchange / precipitation (using NaCl):
H3AlF6 + 3 NaCl → Na3AlF6↓ + 3 HCl
Alternatively using soda ash (Na₂CO₃):
H3AlF6 +3/2 Na2CO3 → Na3AlF6↓ + 3/2 CO2 + 3/2 H2O
The double-replacement reaction drives Na₃AlF₆ precipitation as a sparingly soluble solid, while the by-product (HCl or CO₂/H₂O) remains in the liquid phase and is separated.
Step-by-Step Process Description (PFD Basis)
Equipment list is presented in the Appendix
Unit 1 — HF Absorption & Feed Preparation
- Anhydrous or aqueous HF is absorbed in deionised water to produce dilute HF solution (typically 10–20 wt%)
- Al(OH)₃ (aluminium trihydrate, ATH) is slurried in water and fed as a controlled suspension
- Operating conditions: ambient temperature (20–30°C); atmospheric pressure
- Key parameter: stoichiometric HF:Al(OH)₃ molar ratio = 6:1 (slight HF excess of 2–5% typically applied to ensure complete aluminium complexation)
Unit 2 — Reaction Vessel (Complexation)
- HF solution and Al(OH)₃ slurry are contacted in a stirred tank reactor (STR) or series of reactors
- H₃AlF₆ solution forms as a clear, colourless to slightly yellow acidic liquor
- Temperature: 40–70°C (exothermic — cooling required)
- Pressure: atmospheric
- Residence time: 30–90 minutes
- pH: 2–4 (strongly acidic)
- Key parameter: complete dissolution of Al(OH)₃ confirmed by clarity of solution and Al assay
Unit 3 — Precipitation Reactor
- H₃AlF₆ solution is transferred to a precipitation STR where sodium chloride solution (brine) or soda ash solution is added under controlled addition rate
- Na₃AlF₆ precipitates immediately as a fine white solid
- Temperature: 50–80°C (elevated temperature improves crystal growth and filterability)
- Pressure: atmospheric
- pH: adjusted to 6.5–7.5 to maximise precipitation yield and minimise fluoride in mother liquor
- Residence time: 60–120 minutes
- Key parameter: Na:Al molar ratio (cryolite ratio, CR) controlled precisely at 2.9–3.1 by NaCl/Na₂CO₃ addition rate
Unit 4 — Solid-Liquid Separation
- Cryolite slurry is fed to a vacuum belt filter, rotary drum filter, or centrifuge
- Mother liquor (NaCl/HCl or Na₂CO₃/CO₂ solution) is separated and treated for fluoride recovery or recycling
- Filter cake is washed with deionised water to remove residual sodium chloride and impurities
- Cake moisture: 15–25% after filtration
Unit 5 — Calcination (Rotary Kiln)
- Washed filter cake is fed to an internally heated rotary kiln
- Residual moisture and traces of volatile impurities are removed by controlled heating
- Temperature: 300–500°C (sufficient to drive off moisture and improve flowability without sintering)
- Residence time: 30–60 minutes
- Product: pale white to slightly pink granules (pink tint characteristic of Fluorsid product from NaCl route)
Unit 6 — Milling & Classification
- Calcined granular cryolite is milled in a rotary or ball mill to produce milled/powdered grades
- Classified by particle size to meet customer specifications (granular: 0.5–3 mm; powder: d₅₀ < 100 μm)
- Final product is bagged in 25 kg PP woven bags or shipped in bulk
Unit 7 — Effluent Treatment
- Mother liquor and wash water are treated for fluoride recovery by precipitation as CaF₂ (using lime) or recycled to the HF absorption step
- HCl by-product (NaCl route) may be recovered for sale or neutralised
- Strict fluoride discharge limits apply in all jurisdictions (typically <2–10 mg F⁻/L in effluent)
Process Efficiency
| Parameter |
Value |
| Fluorine yield (HF → Na₃AlF₆) |
88–95% |
| Aluminium yield (Al(OH)₃ → Na₃AlF₆) |
92–97% |
| Na₃AlF₆ purity |
>98% (anode-grade); >99.5% (premium) |
| Cryolite Ratio (CR = Na:Al) |
2.9–3.1 (target 3.0) |
| Moisture (final product) |
<0.5% |
| Sulphate content |
<0.3% |
| SiO₂ content |
<0.1% |
| Fe₂O₃ content |
<0.05% |
Economic Performance
| Parameter |
Indicative Value |
| Parameter |
Indicative Value |
| Raw material costs |
~60–70% of production cost (HF dominant) |
| HF consumption |
~1.05–1.10 kg HF per kg Na₃AlF₆ (theoretical: 1.03) |
| Al(OH)₃ consumption |
~0.36–0.38 kg per kg Na₃AlF₆ |
| NaCl consumption |
~0.50–0.55 kg per kg Na₃AlF₆ (NaCl route) |
| Energy consumption |
~0.8–1.2 GJ/tonne Na₃AlF₆ (mainly for calcination) |
| Market price (2024) |
USD 600–1,200 per tonne (granular/milled) |
The HF route is the most cost-effective at large scale (>10,000 t/yr capacity) due to well-established supply chains for HF and Al(OH)₃, and high product purity that commands premium pricing from aluminium smelters.
Known Deployments & Producers
| Producer |
Country |
Technology Basis |
Capacity |
| Fluorsid |
Italy (Cagliari, Sardinia) |
HF / Al(OH)₃ / NaCl route |
Major European
producer |
| Mexichem (Orbia) |
Mexico /
Global |
HF-based |
Integrated
fluorochemical group |
Jinhe
Chemicals |
China
(Henan) |
HF-based |
~100,000 t/yr
capacity |
| Do-Fluoride Chemicals |
China |
HF-based |
Major Chinese
producer |
| Sinochem Lantian |
China |
HF-based |
Integrated
fluorine chemicals |
Navin
Fluorine |
India |
HF-based |
Leading Indian fluorochemicals producer |
China dominates global synthetic cryolite production with numerous producers, primarily operating the HF/Al(OH)₃ route integrated with domestic HF production from fluorspar.
Licensors
Unlike major petrochemical processes, synthetic cryolite production technology is not widely licensed on a commercial basis through formal licensing programmes — most producers operate proprietary process designs developed in-house or acquired through technology transfer. There are no widely published dedicated technology licensors for this process in the same manner as refinery or petrochemical technologies. Process engineering firms such as Outotec (now Metso Outotec), Prayon, and fluorochemical specialists have provided engineering services for cryolite plant design, but formal licensing agreements and royalty-based technology packages are not the norm in this industry segment.
References
- Fluorsid (Nov 5, 2019). Synthetic Cryolite
- Ataman Chemicals. Synthetic Cryolite (Accessed Mar 5, 2026)
- Safiev Kh., Naimov N.A., Ruziev J.R., Akhmadshoev I.Sh., Juraqulov A.M., Murodiyon A., Nemchinova N.V. Physicochemical parameters of a hydrochemical technology employing sodium chloride to obtain cryolite used in aluminium production. iPolytech Journal. 2022;26(2):348-356. DOI: 10.21285/1814-3520-2022-2-348-356
- Wan B., Li W., Sun W., Liu F., Chen B., Xu S., Chen W., Yi A. Synthesis of Cryolite (Na3AlF6) from Secondary Aluminum Dross Generated in the Aluminum Recycling Process. Materials (Basel). 2020 Sep 2;13(17):3871. DOI: 10.3390/ma13173871. PMID: 32887240; PMCID: PMC7503661
- Henan Jinhe Industry Company. High Purity Synthetic Cryolite for Aluminium Smelting (Accessed Mar 5, 2026)
- 24 Chemical Research (Dec 4, 2025). Report 24CR-227592: Synthetic Cryolite Market, Global Outlook and Forecast 2025-2032
- Wikipedia. Cryolite (Page version: Feb 22, 2026)
Unit 1 — HF Absorption & Feed Preparation
- HF storage tank — Monel or HDPE-lined, pressure-rated, with double containment bund; stores anhydrous or aqueous HF feed
- HF absorption column — packed column (PTFE or PVC internals) for dissolving anhydrous HF gas into deionised water to target concentration (10–20 wt%)
- Deionised water system — ion exchange resin unit + reverse osmosis membrane skid
- Al(OH)₃ storage silo — carbon steel with fluidisation pads and dust filter
- Al(OH)₃ slurry preparation tank — agitated HDPE or rubber-lined carbon steel vessel with high-shear impeller
- Slurry feed pump — peristaltic or centrifugal (rubber-lined), for Al(OH)₃ suspension transfer
- HF metering pump — diaphragm pump (PTFE wetted parts), flow-controlled with mass flow meter (Coriolis)
- Acid-resistant instrumentation — pH probes (PTFE body), conductivity sensors, temperature transmitters (all HF-resistant materials)
Unit 2 — Complexation Reactor
- Stirred tank reactor (STR) ×2 in series — polypropylene-lined or HDPE-lined carbon steel; turbine or anchor impeller; 10–50 m³ working volume per vessel
- Reactor jacket / cooling coils — cooling water system to manage exothermic heat of reaction; PTFE-lined coils or external heat exchanger (graphite block or PTFE shell-and-tube)
- HF scrubber / vent treatment — packed bed scrubber (caustic soda or lime solution) on reactor vent to capture HF vapour emissions
- Reflux condenser — PTFE or fluoropolymer-lined, mounted on reactor vent line
- In-line Al assay analyser — ICP-OES feed loop or manual sampling point for dissolved Al verification
- Temperature & pH control loop — PID-controlled with automated HF/Al(OH)₃ feed ratio adjustment
Unit 3 — Precipitation Reactor
- Stirred precipitation tank ×2–3 — HDPE-lined or rubber-lined carbon steel; 20–100 m³; slow-speed anchor or gate impeller to promote crystal growth
- NaCl brine preparation tank — agitated carbon steel vessel with saturated NaCl solution feed (alternatively Na₂CO₃ dosing tank)
- Brine / soda ash metering pump — diaphragm metering pump with flow control (controls CR = Na:Al ratio)
- pH controller — automated dosing of NaOH or HF trim solution to maintain pH 6.5–7.5
- Crystal size analyser — inline laser diffraction particle size probe (e.g. Malvern Parsum) for crystal morphology monitoring
- Heat exchanger — shell-and-tube (graphite or PTFE) for heating precipitation slurry to 50–80°C
Unit 4 — Solid-Liquid Separation
- Vacuum belt filter or rotary drum filter — rubber-lined drum or belt; polypropylene filter cloth; continuous operation; primary workhorse for large-scale plants
- Centrifuge (peeler or pusher type) — polypropylene-lined bowl for higher-throughput or where purer filter cake is required
- Wash water distribution system — spray nozzle array for counter-current cake washing on belt filter
- Mother liquor collection tank — HDPE-lined; receives filtrate for fluoride recovery or recycle
- Vacuum pump / vacuum receiver — liquid ring vacuum pump (PTFE or stainless); stainless steel receiver vessel
- Filter cloth cleaning system — automatic spray wash bar for cloth regeneration
Unit 5 — Calcination
- Rotary kiln — carbon steel shell, refractory-lined; direct or indirect gas/oil/electric fired; 2–4 m diameter × 10–20 m length; typically natural gas or LPG fired
- Kiln feed conveyor — stainless steel screw conveyor or belt conveyor from filter to kiln feed hopper
- Kiln exhaust gas treatment — baghouse filter (polyester or PTFE bags) + wet scrubber for particulate and HF/fluoride vapour capture
- Rotary cooler — directly after kiln discharge; air-cooled drum cooler or fluidised bed cooler to reduce product temperature before milling
- Thermocouple array — multiple K-type or S-type thermocouples along kiln length for temperature profiling
Unit 6 — Milling & Classification
- Rotary impact mill or ball mill — for coarse-to-medium grinding of calcined granules; stainless steel or ceramic-lined
- Air classifier — for particle size separation (d₅₀ control); produces granular and milled product grades simultaneously
- Cyclone separator — for fine particle recovery from classifier airstream
- Product silo ×2 — carbon steel with fluidisation pads; separate silos for granular and milled grades
- Bagging machine / bulk loader — automatic 25 kg PP bag filling station with dust extraction hood; bulk truck/container loading spout
Unit 7 — Effluent Treatment
- Fluoride precipitation tank — agitated rubber-lined vessel; lime (Ca(OH)₂) slurry addition to precipitate CaF₂ from mother liquor and washwater
- Lime slurry preparation system — slaker + agitated lime slurry tank
- Lamella clarifier or thickener — for CaF₂ sludge settling and mother liquor clarification
- Filter press — for CaF₂ sludge dewatering (recoverable as low-grade fluorspar by-product)
- Effluent polishing tank — final pH adjustment (NaOH/H₂SO₄) before discharge
- Online fluoride analyser — ISE (ion selective electrode) or photometric analyser on final effluent stream for continuous compliance monitoring
- HCl recovery absorber (NaCl route only) — packed absorption column to recover HCl by-product as aqueous hydrochloric acid for sale or neutralisation
Utilities & Safety Infrastructure
- HF emergency scrubbing system — caustic spray deluge system covering all HF handling areas; standalone backup power supply
- HF personal protection stations — calcium gluconate emergency wash units at all HF exposure points
- Gas detection system — fixed HF gas detectors (electrochemical sensors) throughout plant with automatic alarm and shutdown interlock
- Deionised water system — multi-stage RO + ion exchange for process water quality control
- Instrument air compressor & dryer — oil-free compressor + refrigerant/adsorption dryer for pneumatic instruments and valve actuators
- DCS / SCADA system — distributed control system for full plant automation, interlocks, and recipe management
Materials of Construction Summary
| Service |
Material |
| HF contact (liquid) |
HDPE, PTFE, Monel, Hastelloy C |
| H₃AlF₆ solution |
HDPE, rubber-lined steel, PP |
| Na₃AlF₆ slurry |
Rubber-lined steel, PP |
| Calcination (high temp) |
Carbon steel + refractory lining |
| Milling |
Stainless steel 316L or ceramic |
| Effluent treatment |
Rubber-lined steel, FRP |