Product
- Product
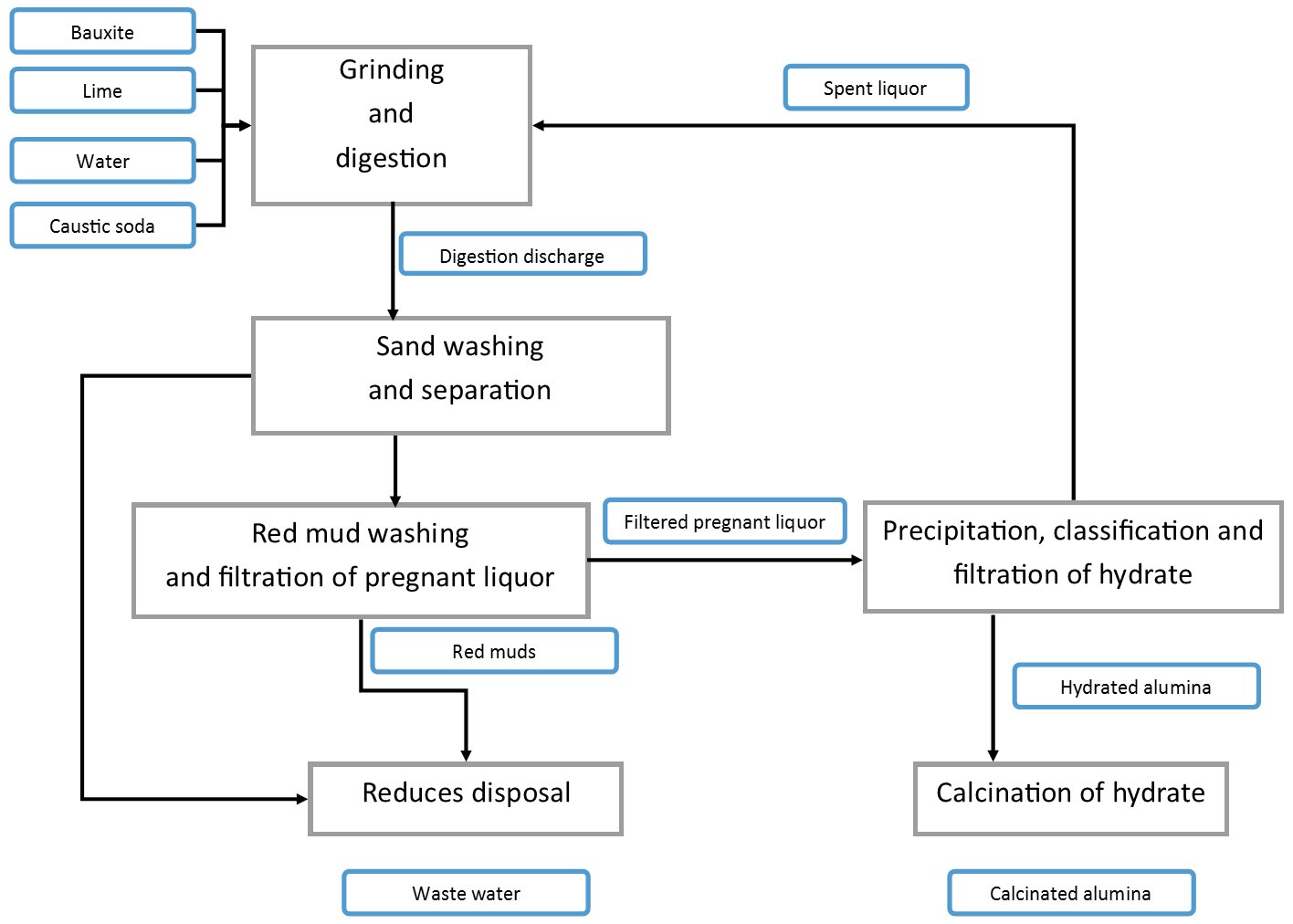
- Spent Caustic Liquor
- Names
- Spent Bayer Liquor; Depleted Aluminate Liquor
-

- #PS756
- Main Product
- Sodium Hydroxide
- Segment
- Chemicals
- Main-Family
- Inorganics
- Sub-Family
- Inorganic Hydroxides
- Physical State
-
Solid
Description
Your insights will be shown here
Product Communicator
| Title | Date | |
|---|---|---|

|
2/27/2026 |
Identifiers
No Identifiers defined
Chemical Data
- Specific Gravity
- 1.00
Crude Data
- API Gravity
- 10
- Country
Product Settings
- Default
- Status
- A
Content provided by
| Transaction | Name | Date |
|---|---|---|
| Modified by |
|
2/26/2026 8:49 PM |
| Added by |
|
2/26/2026 8:34 PM |