Product
- Product
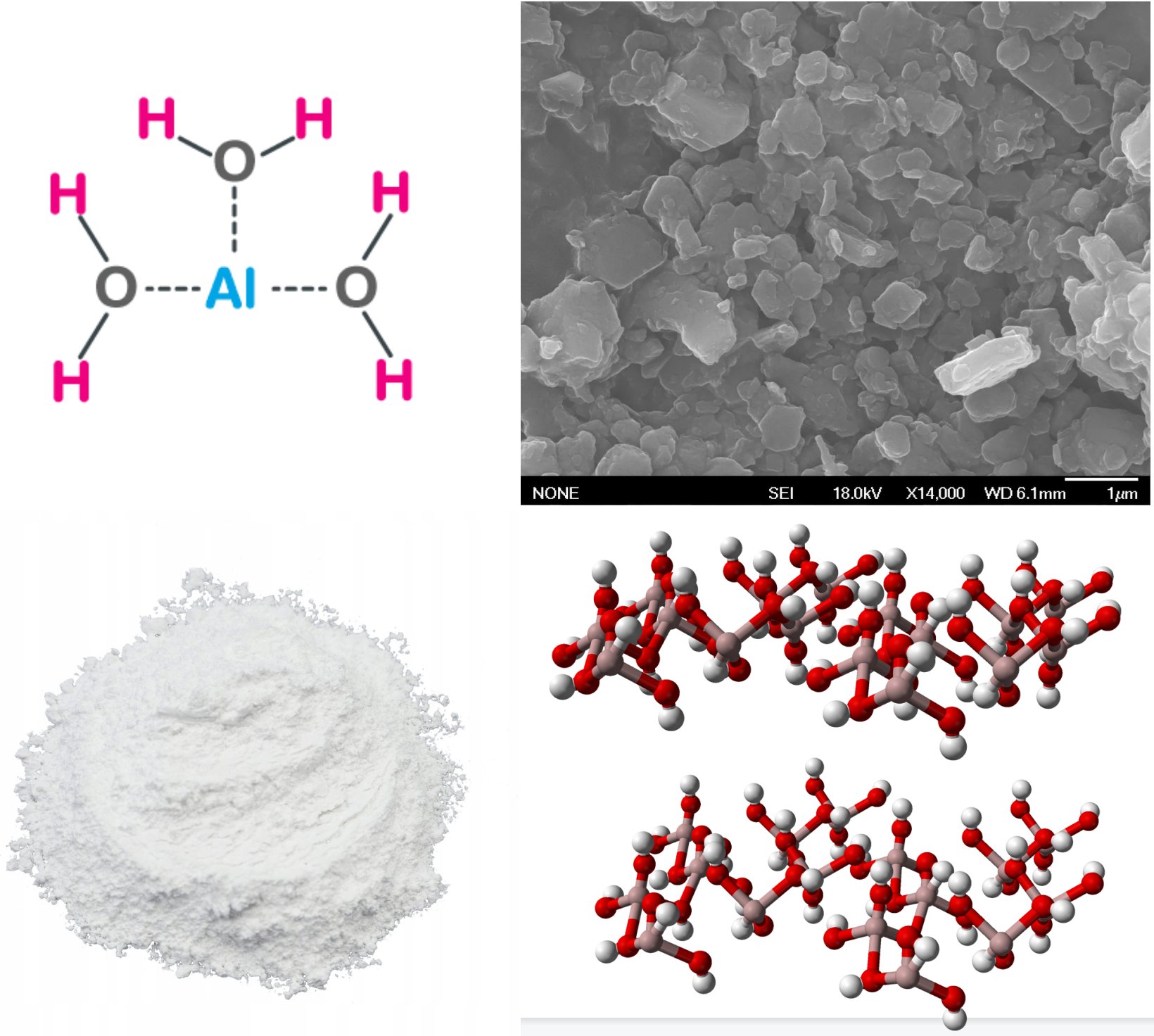
- Aluminium Trihydroxide
- Names
- Aluminium Hydroxide; Trihydroxidoaluminium; Aluminic acid; Alumanetriol; Aluminium(III) Hydroxide; Aluminic Hydroxide; Aluminium Trihydroxide; Hydrated Alumina; Orthoaluminic Acid
-

- #PS754
- Main Product
- Aluminium Hydroxides
- Segment
- Chemicals
- Main-Family
- Inorganics
- Sub-Family
- Inorganic Hydroxides
- Physical State
-
Solid
Description
Your insights will be shown here
Product Communicator
| Title | Date | |
|---|---|---|

|
2/26/2026 |
Identifiers
-
 CAS Number
CAS Number
- 21645-51-2
-
 EC Number
EC Number
- 244-492-7
-
 ECHA InfoCard
ECHA InfoCard
- 100.040.433
-
 IUPAC Name
IUPAC Name
- Aluminium Hydroxide
-
 PubChem ID
PubChem ID
- 10176082
Chemical Data
- Chemical Formula
-
Al(OH)3
- Molecular Weight (g/mol)
- 78
- Sulfur Content (wt%)
- 0
- Specific Gravity
- 2.42
Crude Data
- API Gravity
- -73.03
- Country
Product Settings
- Default
- Status
- A
Content provided by
| Transaction | Name | Date |
|---|---|---|
| Modified by |
|
2/26/2026 1:24 PM |
| Added by |
|
2/26/2026 12:27 PM |